MNCNH Portfolio
Abbreviation |
Definition |
|---|---|
ACMR |
All-Cause Mortality Rate |
ACS |
Antenatal Corticosteroids |
AI ultrasound |
Artificial Intelligence assisted ultrasound |
ANC |
Antenatal Care |
ASFR |
Age-Specific Fertility Rate |
BEmONC |
Basic Emergency Obstetric and Newborn Care |
CEmONC |
Comprehensive Emergency Obstetric and Newborn Care |
CPAP |
Continuous Positive Airway Pressure |
CSMR |
Cause-Specific Mortality Rate |
ENN |
Early Neonatal |
GBD |
Global Burden of Disease |
IFA |
Iron and Folic Acid |
IFD |
In-Facility Delivery |
IV iron |
Intravenous iron |
LBW |
Low Birth Weight |
LBWSG |
Low Birth Weight and Short Gestation |
LNN |
Late Neonatal |
MMS |
Multiple Micronutrient Supplements |
MNCNH |
Maternal, Newborn, and Child Nutrition and Health |
OL |
Obstructed Labor |
PAF |
Population Attributable Fraction |
PPD |
Postpartum Depression |
PTB |
Preterm Birth |
RDS |
Respiratory Distress Syndrome |
RR |
Relative Risk |
RT |
Research Team |
SBR |
Stillbirth (to live birth) Ratio |
V&V |
Verification and Validation |
WRA |
Women of Reproductive Age |
YLDs |
Years Lived with Disability |
YLLs |
Years of Life Lost |
1.0 Overview
This document is the overall page for the Maternal, Newborn, and Child Nutrition and Health (MNCNH) Portfolio simulation and contains information that relates to all modeled subcomponents included in the simulation.
2.0 Modeling aims and objectives
This simulation aims to answer the research question: What impacts would different (combinations of) interventions have on maternal, newborn, and child nutrition and health outcomes? We model three locations currently: Nigeria, Ethiopia, and Pakistan. We aim to incorporate quite an expansive list of interventions in various stages of development that are under consideration by the Gates Foundation. The “combinations of” part of the research question is crucial – interventions might interact, for example due to prevention reducing the need for treatment down the line.
The MNCNH Portfolio simulation builds on work our team has done in other simulations of pregnancy and early childhood. The most recent was the Nutrition Optimization (NO) simulation, which (as the name suggests) focused particularly on nutrition interventions. That simulation allowed us to estimate the impacts of each intervention, and crucially also how the interventions interacted. “Optimization” refers to the fact that we used the output of the NO sim to calculate optimal allocation of money to have the biggest impact given a budget, taking all these interactions into account.
Nutrition interventions continue to be included in the MNCNH portfolio sim, but the “portfolio” is broader, including non-nutrition interventions as well. As before, we also plan to estimate costs and calculate optimal budget allocation, possibly with improved costing methodology. This page serves as documentation for the simulation part of the project, which is focused on estimating burden under a variety of scenarios designed to surface all the relevant product interactions.
Simulating more interventions means simulating more risks and causes for those interventions to act on, simulating more details of the healthcare system to model how those interventions would be delivered, and including more detail in the intrapartum (labor and delivery) and neonatal time periods.
We plan to complete this work in 3 waves.
Wave 1 will include the basic model design, outlines of the healthcare system, and some interventions (AI ultrasound, RDS management).
Wave 2 will add in some antenatal supplements (MMS, IV iron), the hemoglobin risk for birthing parents, all downstream causes affected by hemoglobin, and higher level delivery facility interventions.
Wave 3 will add in gestational blood pressure and relevant causes and risks including pre-eclampsia care and downstream effects of high blood pressure.
As of February 2026, Wave 1 is complete in both documentation and implementation and Wave 2 is mostly documented and partly implemented.
3.0 Concept model diagram and submodels
As in the NO simulation, rather than simulate an entire population of all ages and sexes, this simulation includes only pregnant people and the neonates they give birth to. We start the simulation with a cohort of simulants all at the beginning of pregnancy, and move them in lockstep through their pregnancies. For those pregnancies that result in a live birth, we then simulate a neonate (we do not model twins) through the first month of life. In this way, our simulation represents all the people who may benefit from the interventions of interest, without wasting computational resources on simulating irrelevant people, such as adult males. We call the potential simulant pair we follow through the simulation (pregnant person and neonate) a “simulant dyad.”
This model is different than NO and other simulations we’ve done in that it follows a decision-tree-like format in which we jump directly to from one decision point to the next rather than taking equal-sized steps through time. For this reason, throughout this model we calculate and express events in terms of probabilities, rather than rates per person-time or similar.
In part because of this unusual handling of time, this model also contains much more “fortune-telling” than a typical sim: instances in which the order that decisions are made in the simulation does not correspond to the real-life order in which events occur. For example, one of the first attributes assigned to each pregnant person, in the initial attributes module, is their broad pregnancy outcome: whether their pregnancy will result in a live/stillbirth, or an abortion/miscarriage/ectopic pregnancy. In real life, however, this doesn’t all get determined at the same time. Whether a pregnancy is ectopic, for example, is determined at the time of implantation (around 3-4 weeks gestation). A full-term pregnancy and one that will end in miscarriage are indistinguishable until the miscarriage occurs, which is much later (up to 24 weeks gestation). Our simulation makes all of these decisions at once by choosing broad pregnancy outcome. The broad pregnancy outcome then informs the attribute of whether and when the pregnant person will attend antenatal care (ANC). This leads to the strange situation that, in our simulation, simulants attending early ANC are already “fated” to have, or not have, a miscarriage, hence the term “fortune-telling.” While odd to think through, this is a valid approach as long as we don’t need to simulate a causal relationship between broad pregnancy outcome and ANC attendance. We have to simulate causal relationships for each step in the pathways between our interventions and outcomes, and these have to be in temporal order; otherwise something an intervention will avert from happening could already have occurred before the intervention has a chance to act. For all relationships between attributes that aren’t in a pathway between an intervention and an outcome, however, the order doesn’t matter. We can choose however is convenient, and often make the choice based on the data that are available, though we should keep in mind that “fortune-telling” does make the model a bit harder to reason about and less intuitive. In the case of broad pregnancy outcome and ANC attendance, we currently don’t model interventions that impact either of these attributes. Even in the future if we modeled an intervention that increased ANC attendance, that intervention wouldn’t act through changing broad pregnancy outcome, hence there is no need to model a causal relationship.
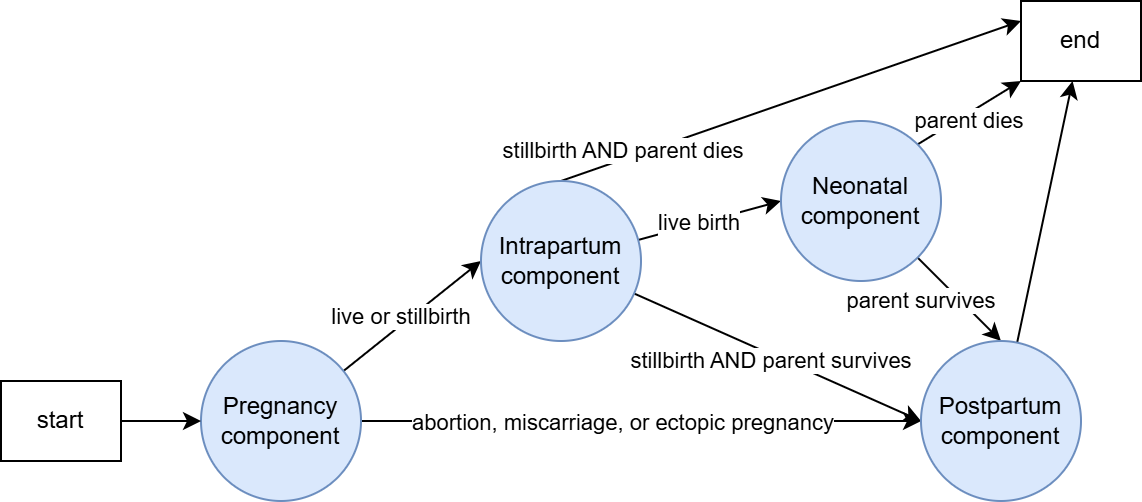
The overall simulation model is divided into four “components,” which are differentiated by the timespan and the simulant that they model.
The Pregnancy component, which models from the beginning of pregnancy until the start of labor.
The Intrapartum component, which models labor and delivery, including delivery complications.
The Neonatal component, which models the first month of life for newborns.
The Postpartum component, which models the six weeks after the end of pregnancy for the pregnant person.
Warning
When we say “component” here, we mean something distinct from a Vivarium component.
Graphically, the component breakdown looks like this:

Note
Do not interpret the x-axis in this diagram as time, since e.g. the duration of pregnancy is not at all constant. Also, if misinterpreted this way, the x-axis would be wildly not to scale.
However, the only situation in which all components are actually reached for a given simulant dyad is the case in which the pregnancy results in a live birth and the birthing person survives childbirth. In other situations, some components will not be reached. The rules by which components flow into other components are as follows:
All simulant dyads start at the pregnancy component.
If the birth outcome from the pregnancy component is a live or stillbirth (NOT abortion/miscarriage/ectopic pregnancy), proceed to the intrapartum component. Otherwise, skip to the postpartum component.
At the end of the intrapartum component, if the birth outcome from the pregnancy component is a live birth, proceed to the neonatal component. Otherwise, if the birth parent survives childbirth, proceed to the postpartum component.
At the end of the neonatal component, if the birth parent survived childbirth in the intrapartum component, proceed to the postpartum component.
Here is a graphic representation of the same information:
Each component is further subdivided into “modules,” which are organized by topic (rather than by time/simulant as in the components). Each module may have some simulant dyad attributes as input (values it needs) and some simulant dyad attributes as output (values it initializes). Module outputs may be used as inputs to other modules and/or serve as information for verification and validation and/or simulation results. For clarity, in the tables below we will write the modules in an order that satisfies the following property: each variable is defined as a module output prior to being used as a module input. This helps us make sure we aren’t creating any cyclic dependencies. Technically, any order satisfying this property is an equivalent, valid order in which the modules could be run in the simulation.
Note
There is a template to use when creating new module pages.
Pregnancy component
Module |
Inputs |
Outputs |
Nested subcomponents |
|---|---|---|---|
|
|||
|
|
||
|
|
||
|
|
||
|
|
||
|
|
||
|
|
||
|
|
Intrapartum component
Note
Only live births or stillbirths (NOT abortions/miscarriages/ectopic pregnancies) will proceed to the intrapartum component, as described above. Both antepartum and intrapartum stillbirths will proceed to the intrapartum component. However, antepartum stillbirths will only be eligible for intrapartum interventions that act on maternal health (such as misoprostol and azithromycin) and will not be eligible for intrapartum interventions intended for neonatal health (such as antenatal corticosteroids) as the fetus will have already passed prior to the onset of labor, but delivery of the fetal remains will still be necessary. Intrapartum stillbirths will remain eligible for all intrapartum interventions.
Warning
As currently designed, the intrapartum component models an intervention for misoprostol to prevent postpartum hemorrhage (PPH) in home birth settings only. We do not consider any interventions for PPH prevention at facility settings nor do we model the expected greater incidence of PPH in home settings relative to facility settings.
Therefore, as written, the incidence of PPH by delivery setting will be miscalibrated to the expectation in reality. We plan to continue with the implementing the model as written while noting this limitation until we implement a strategy to address this (see related ticket here)
Module |
Inputs |
Outputs |
Nested subcomponents |
|---|---|---|---|
|
|
||
|
|
||
|
|
Neonatal component
Note
Only live births proceed to the neonatal component, as described above.
Module |
Inputs |
Outputs |
Nested subcomponents |
|---|---|---|---|
|
|
Postpartum component
Module |
Inputs |
Outputs |
Nested subcomponents |
|---|---|---|---|
|
|
||
|
|
||
|
|
Concept Model Map:
Please note that this is designed as a helpful visual aid, but is not designed to be fully inclusive of all work or modeled components.
Pregnancy, influenced by ANC attendance
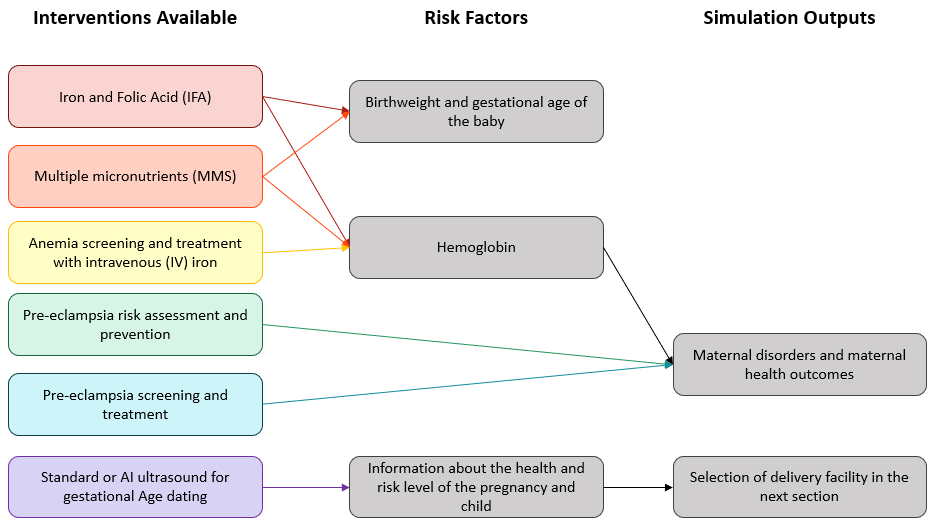
Intrapartum, influenced by delivery facility
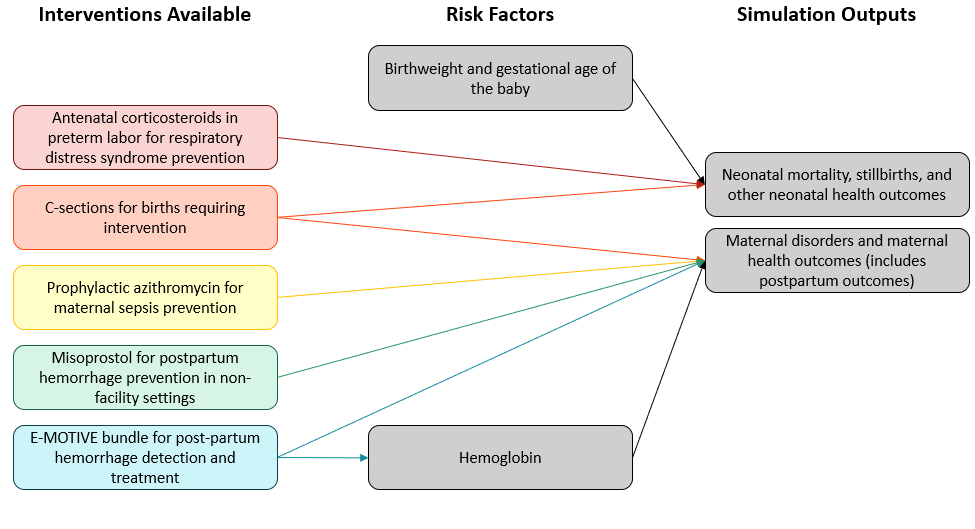
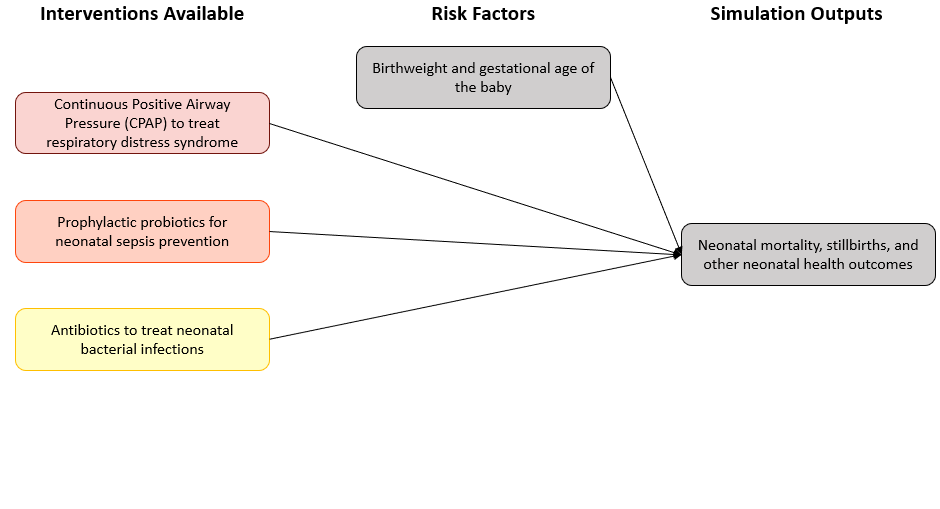
Neonatal, influenced by neonatal care facility
3.1 Scenario information
Note
Scenarios were reworked for model version 18.4. To see the definition of scenarios used for prior models, see the record in this pull request. Note that scenario design for this simulation is expected to undergo an additional future rework to achieve compatibility with an “emulator” design.
Scenario |
Ultrasound coverage |
Ultrasound type |
Oral iron coverage |
Hemoglobin screening coverage |
Ferritin screening coverage |
IV iron coverage |
Note |
|---|---|---|---|---|---|---|---|
|
Defined in the baseline coverage section of the AI ultrasound module page |
Defined in the baseline coverage section of the AI ultrasound module page |
Defined in the baseline coverage section of the oral iron supplementation page (use the |
Defined in the baseline coverage section of the anemia screening intervention page |
Defined in the baseline coverage section of the anemia screening intervention page |
Defined in the baseline coverage section of the IV iron page |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
100% |
100% AI-assisted |
Baseline |
Baseline |
Baseline |
Baseline |
When compared to 2, shows how AI ultrasound–>facility choice pathway can improve outcomes by having more preterm deliveries deliver in facility settings |
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
100% at ANC (no ultrasound among those who do not attend ANC) |
100% AI-assisted |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
100% at ANC (no ultrasound among those who do not attend ANC) |
100% standard |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
100% at ANC (no ultrasound among those who do not attend ANC) |
100% AI-assisted |
Baseline |
Baseline |
Baseline |
Baseline |
Not including anemia related interventions in this run as implementation of all model components is incomplete as of October 2025 |
|
Baseline |
Baseline |
100% MMS |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
100% |
100% |
100% |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
100% at ANC (no ultrasound among those who do not attend ANC) |
50% standard US, 50% AI-assisted US |
Baseline |
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
100% of eligible population |
100% of eligible population |
Baseline |
Scenario |
Azithromycin coverage |
Corticosteroid coverage |
Misoprostol coverage |
Note |
|---|---|---|---|---|
|
Defined on intrapartum intervention model document |
Defined on intrapartum intervention model document |
Defined on intrapartum intervention model document |
|
|
Baseline |
100% at BEmONC and CEmONC, baseline at home |
Baseline |
|
|
Baseline |
100% at BEmONC and CEmONC, baseline at home |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
100% at BEmONC and CEmONC, baseline at home |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
100% at BEmONC and CEmONC, baseline at home |
100% at BEmONC and CEmONC, baseline at home |
Baseline |
Not including misoprostol scale-up in this run because as of October 2025 we have known calibration issues with our hemorrhage model by delivery facility setting |
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
50% at BEmONC and CEmONC, baseline at home |
0% |
0% |
|
|
0% |
0% |
50% among eligible population (attends ANC and delivers at home) |
|
|
Baseline |
0% coverage at all delivery location types |
Baseline |
see neonatal table for CPAP coverage |
|
Baseline |
100% coverage at BEmONC and CEmONC facilities, baseline at home |
Baseline |
see neonatal table for CPAP coverage |
|
Baseline |
100% coverage at BEmONC and CEmONC facilities, baseline at home |
Baseline |
see neonatal table for CPAP coverage |
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
Scenario |
CPAP coverage |
Antibiotics coverage |
Probiotics coverage |
Note |
|---|---|---|---|---|
|
Defined on the CPAP intervention model document |
Defined on the neonatal antibiotic intervention document |
Defined on the probiotics intervention model document |
Baseline coverage values are delivery facility-specific |
|
100% at BEMONC and CEMONC, baseline at home |
Baseline |
Baseline |
|
|
100% at BEMONC and CEMONC, baseline at home |
Baseline |
Baseline |
|
|
Baseline |
100% coverage |
Baseline |
|
|
Baseline |
Baseline |
100% at CEMONC and BEMONC, baseline at home |
|
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
100% at BEMONC and CEMONC, baseline at home |
100% |
100% at BEMONC and CEMONC, baseline at home |
|
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
|
|
100% coverage at BEmONC and CEmONC facilities, baseline at home |
Baseline |
Baseline |
See intrapartum table for ACS coverage |
|
100% coverage at BEmONC and CEmONC facilities, baseline at home |
Baseline |
Baseline |
See intrapartum table for ACS coverage |
|
0% coverage at all delivery location types |
Baseline |
Baseline |
See intrapartum table for ACS coverage |
|
Baseline |
Baseline |
Baseline |
|
|
Baseline |
Baseline |
Baseline |
4.0 Outputs/Observers
Specific observer outputs and their stratifications may vary by model run as needs change. Modifications to default will be noted in the model run requests tables. Note that the observers and outputs listed here are different from the module outputs above. The outputs of the module are intended to be intermediate values that may or may not be included as observed simulated outputs.
Default stratifications to all observers should include scenario and input draw.
Note
Observers cannot support more than 15 stratifications. Design of simulation observers should take this into account.
Observer |
Default stratifications |
Note |
|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
Included. Confirm this represents “eligible birth counts”? |
|
|
Included. Confirm this represents “eligible birth counts”? |
|
|
Included. Confirm this represents “eligible birth counts”? |
7a. Maternal population counts: hemoglobin-related parameters |
|
|
7b. Maternal population counts: other parameters |
|
|
|
|
Observe the following statistics about the \(\text{ACMRisk}_i\) value described on the neonatal mortality page,
which is implemented as the
All of these quantities can aggregate across seeds in the normal way (summation). |
|
|
Observe the following statistics about the \(\text{CSMRisk}^k_i\) value described on the neonatal mortality page,
which is implemented as the
All of these quantities can aggregate across seeds in the normal way (summation). |
|
|
For each living simulant, take the modeled-cause CSMRisks (same pipelines as used in the previous observer), divide them each by the ACMRisk (same pipeline as observer #8), sum them and then subtract 1. If negative, clip this value to zero. This emulates the factor by which the modeled-cause CSMRisk exceeds ACMRisk, requiring a hack to prevent other-causes CSMRisk from being negative. Then observe:
All of these quantities can aggregate across seeds in the normal way (summation). |
|
|
Inclusive of anemia YLDs accrued during pregnancy and the postpartum period |
|
|
Inclusive of anemia person-time accrued during pregnancy and the postpartum period |
Todo
Determine whether we want to continue to have duplicate information like:
Stratifying the birth observer by neonatal interventions,
AND separately observing neonatal intervention counts
5.0 Model runs
Parameter |
Value |
Note |
|---|---|---|
Location(s) |
|
|
Number of draws |
10 |
See next row for which specific draws to be used. Based on calculations from the Nutrition Optimization project: production run number divided in half for default V&V runs |
Draw numbers |
115, 60, 118, 197, 79, 244, 22, 167, 146, 71, 28, 156, 94, 170, 109, 26, 35, 114, 178, 127 |
The standard number of draws available for most of our model input parameters is 500 for GBD 2021 and 250 for GBD 2023. There are a few instances where our input data have different numbers of draws available. Those instances are summarized in the table following this one. To account for the varying number of draws available for our input data, we will pre-specify which draws to select according to the numbers listed here. 20 draws have been listed although the default number of draws for V&V model runs is 10 - the first 10 numbers in this list should be used for V&V runs. This list of numbers was first generated for GBD 2021 data starting in model 11.0 by sampling a random number between 0 and 499 and resampling when a number was generated that had the same remainder after dividing by 100 or 250 as a number that was already in the list. This strategy ensures that we do not run multiple draws that have identical data for any parameter in our model. These draws included: [115, 60, 368, 197, 79, 244, 272, 167, 146, 71, 278, 406, 94, 420, 109, 26, 35, 114, 428, 218]. The final number in the list (218) was updated from (170) in September of 2025 to account for new input data with 250 draws. This list was later updated for GBD 2023 data starting in model 19.0 by taking the remainder of each number in the GBD 2021 list after dividing by 250 and re-sampling a new value for 218 (which had a duplicate value % 100 as another number in the list). |
Population size per draw |
200,000 |
Based on calculations from the Nutrition Optimization project |
Randomness key columns |
[‘entrance_time’,’age’] |
Note that each row of the population table in this simulation contains a pregnant simulant AND the outcome of that simulant’s pregnancy. Therefore, the conversion of a stillbirth to a live birth between simulated scenarios in this simulation will not result in a new row added to the simulation state table and therefore will not change the state table index value of other simulants like occured in the IV iron simulation and resulted in disruptions to common random numbers between scenarios. Therefore, these randomness key columns are expected to be sufficient for this simulation. |
Age start (initialization) |
10 |
Applies to pregnant population only |
Age end (initialization) |
54 |
Applies to pregnant population only |
Age start (observation) |
N/A. All pregnant simulants observed from start of pregnancy. All neonatal simulants observed from birth. |
|
Age end (observation) |
N/A; All pregnant simulants observed through conclusion of relevant modeled outcomes. All neonatal simulants observed until 28 days (end of late neonatal age group) |
Pregnant/birthing simulants do not age in this simulation |
Parameter |
Number of draws |
Strategy for GBD 2021 (Models <19.0) |
Strategy for GBD 2023 (Models 19.0+) |
Note/reference |
|---|---|---|---|---|
Standard GBD data |
500 for GBD 2021, 250 for GBD 2023 |
Use 500 draws as is |
Use 250 draws as-is |
|
Hemoglobin risk exposure (using GBD 2023 data) |
100 |
Copy 5 times so that draw 1, 101, 201, 301, and 401 all have the same value, etc. |
Copy 2.5 times so that draw 1, 101, and 201 all have the same value. Note that draws 0-49 will be used three times and draws 50-99 will be used twice. |
|
Hemoglobin risk effects, including those on stillbirth, gestational age, and birthweight that are modeled through the IV iron intervention model |
250 |
Copy twice so that draw 1 and 251 have the same value, etc. |
Use as is |
Note that we have ordered the draws for hemoglobin RRs on gestational age, birth weight, and neonatal sepsis in the same order as we are modeling mediation by gestational age and birthweight in the effect of hemoglobin on neonatal sepsis and therefore expect that these draws will be correlated. |
100 |
Copy 5 times so that draw 1, 101, 201, 301, and 401 all have the same value, etc. |
Copy 2.5 times so that draw 1, 101, and 201 all have the same value. Note that draws 0-49 will be used three times and draws 50-99 will be used twice. |
Note
“Number” is only listed here for models that have already been run. When a model is run, its number is assigned in this excel run tracker. The number will be added to this table when V&V results are posted.
The “blocked by” column only applies to models that have not yet been run. When there is nothing in this column, a model is unblocked and can be implemented. There is no explicit ordering besides this dependency tree for not-yet-run models.
When a model is run and V&Ved successfully, it should be removed from the “blocked by” column for any other model that was blocked by it.
Note
Moving forward, the results for each model should be stored in a subdirectory nested within mnt/team/simulation_science/pub/models/vivarium_gates_mncnh/results/
where the subdirectory name is the model number with “model” prepended to it;
for example, modelX.Y.Z.
The subdirectory names did not match the model numbers before model 6.1, and are given in parentheses next
to the model number for older models.
Number |
Short name |
Description |
Scenarios |
Spec. modifications |
Blocked by |
|---|---|---|---|---|---|
1 ( |
Wave I Pregnancy V&V |
Baseline |
|||
2 ( |
Wave I Maternal disorders V&V |
Baseline |
|||
3 ( |
Wave I Neonatal disorders V&V |
Baseline |
|||
3.1 ( |
Wave I Neonatal disorders V&V with correct LBWSG distribution |
Baseline |
|||
3.2 ( |
Wave I Neonatal disorders V&V with LBWSG component removed |
Baseline |
|||
3.3 ( |
Wave I Neonatal disorders V&V with early NN observer bugfix |
Baseline |
|||
4.1 ( |
Wave I CPAP |
Baseline |
|||
4.2 ( |
Wave I CPAP with observer for counts per facility type |
Baseline |
|||
4.3 ( |
Wave I CPAP with addition of a delivery facility column |
Addition of a delivery facility column in births observer and CPAP availability stratification in neonatal burden observer |
Baseline |
||
4.4 ( |
Wave I CPAP with updated facility determination |
Baseline |
|||
4.5 ( |
Wave I CPAP with mortality bugfix |
Bugfix for negative other causes mortality rates |
Baseline |
||
4.6 ( |
Wave I CPAP with scale-up scenarios |
Baseline and alternative scenarios 2, 3, and 4 |
|||
4.7 ( |
Correct pregnancy duration for abortion/miscarriage/ectopic pregnancies |
Baseline and alternative scenarios 2, 3, and 4 |
|||
5.0 ( |
Wave I neonatal antibiotics with scale-up scenarios |
Baseline and alternative scenarios 2 - 7 |
|||
5.1 ( |
Wave I neonatal antibiotics refactor |
Engineer refactor |
Baseline and alternative scenarios 2 - 7 |
||
6.0 ( |
Wave I neonatal probiotics with scale-up scenarios |
Baseline and alternative scenarios 2 - 10 |
|||
6.0.1 ( |
Wave I neonatal disorders ACMR 200k |
200k population without interventions |
Baseline |
Population increased 10 fold (random seed population size changed from 20k to 200k) |
|
6.0.2 ( |
Wave I neonatal disorders ACMR 2M |
2 million population |
Baseline |
Population increased 100 fold (random seed population size changed from 20k to 2 million) |
|
6.0.3 ( |
Wave I neonatal disorders ACMR with rate conversion |
Rate to probability conversion |
Baseline |
||
6.0.4 ( |
Wave I neonatal disorders ACMR with raw CSMR |
Baseline |
|||
6.1 |
LBWSG PAF changes for Ethiopia |
|
All scenarios |
||
6.2 |
Exponential rate-to-probability conversion |
Same specifications as model 6.1, but this time with the exponential rate-to-probability conversion (\(p= 1 - e^{(-\text{rate} * \text{duration scaling factor})}\)) in this function |
Baseline |
Birth observer updated from output of state table (single row per simulant) to observer detailed in the observer section for all subsequent model runs |
|
6.2.1 |
Fix rate-to-probability equation |
Same as 6.2, but with a fix for this rate to probability equation transcription error (add back in the duration_scaling_factor) and include abortion/miscarriage/ectopic pregnancy fix to birth observer |
Baseline |
||
6.3 |
ENN LBWSG PAF using ENN exposure |
Same specifications as model 6.2 (including the exponential rate-to-probability calculation), but with ENN LBWSG PAF updated to use the ENN LBWSG exposure prevalence rather than the LBWSG exposure at birth |
Baseline |
||
6.4 |
Revert to linear rate-to-probability |
Same specifications as model 6.3 (including the ENN LBWSG PAF using ENN exposure), but with the revision of the rate-to-probability calculation back to \(p = \text{rate} * \text{duration scaling factor}\) |
Baseline |
||
6.5 |
Add maternal population observer |
|
All scenarios |
Maternal population observer added for this run and to be included in all subsequent runs |
|
7.0 |
Wave I neonatal probiotics with effective coverage |
Same as model 6.0 but with effective coverage (only preterm neonates receive probiotics) |
Baseline and alternative scenarios 2 - 10 |
Stratify probiotics observer (#6) with gestational age above/below 37 weeks for V&V |
|
7.0.1 |
Add preterm stratification and fix observers |
Same specifications as 7.0, but with preterm stratification for the probiotics observer included (left out of last run) and fix to the intervention observers to not count stillbirths |
All scenarios |
|
|
7.0.2 |
Update preterm birth parameter |
Update \(p_\text{preterm}\) parameter used in the preterm cause model to use birth exposure rather than age-specific exposure |
All scenarios |
Default |
|
7.1 |
Update neonatal mortality to risks |
Update mortality input data and remove rate to probability conversion: see this PR for full details and accounting of updates. Use the birth LBWSG exposure for calculation of the ENN LBWSG PAF. Use the LNN LBWSG exposure for calculation of the LNN LBWSG PAF. Note that this is incorrect, but an acceptable placeholder until we update in model run 7.2 |
Baseline |
Same modifications as run 7.0.1:
|
|
7.1.1 |
Add CPAP parameter uncertainty |
Add parameter uncertainty interval for CPAP effect size |
All scenarios |
Same as 7.0.1 |
|
8.0 |
Wave I azithromycin |
All scenarios (note new azithromycin scale-up scenario #11) |
Azithromycin stratifications added to observers #1 and #7 (maternal burden and maternal population observers) - to be continued as defaults for all future runs |
||
8.1 |
Cap LBWSG RRs |
|
Baseline |
Same modifications as run 7.0.1 |
|
8.2 |
Update neonatal probiotics effect size |
Update intervention effect size in accordance with line #183 in this PR |
All scenarios |
Same modifications as run 7.0.1 |
|
8.3 |
Update antibiotics modeling strategy |
Update intervention modeling strategy in accordance with this PR |
All scenarios (note that scenarios #6 and #7 have been deleted as they are no longer relevant and scenario #5 no longer has delivery facility-specific coverage) |
Default |
|
9.0 |
Wave I misoprostol |
Baseline and #12 |
Note misoprostol coverage added as a stratifying variable to maternal disorders burden and maternal population observers and delivery facility as a stratifying variable for the maternal disorders burden observer |
||
9.1 |
Antibiotics bugfix for home deliveries |
Bugfix to scale up intervention among home deliveries as well |
All scenarios |
Default |
|
9.2 |
Larger population for obstructed labor V&V |
Larger population size to confirm maternal obstructed labor is not affected by azithromycin |
All scenarios |
10x larger population size (100 seeds of 20_000 population size each = 2_000_000 population size per draw) and 2x as many draws for a total of 20 draws |
|
9.3 |
Additional stratifications for intrapartum V&V |
Updated intervention scenario coverage for intrapartum intervention V&V |
All scenarios – Note changes to scenario numbers 11 and 12 |
|
|
10.0 |
Add postpartum depression |
Postpartum depression added as new maternal disorder cause |
Baseline |
Note that postpartum depression cause should be added to the maternal disorders burden observer |
|
10.1 |
Remove LBWSG effect on neonatal encephalopathy |
Run with no effect between LBWSG risk factor and Neonatal encephalopathy due to birth asphyxia and birth trauma (but keep LBWSG effects on all other outcomes) |
All scenarios |
Default |
|
10.2 |
Same as “Add postpartum depression” with scenario #13 |
Additional scenario #13 (azithromycin results) |
All scenarios |
Default |
|
11.0 |
Add hemoglobin risk exposure model |
Add Hemoglobin risk exposure model. Note that this will be the starting point for the larger wave II hemoglobin module, which will be built out in future model runs |
Baseline |
Default (no new hemoglobin observer required) |
|
11.1 |
VPH LBWSG refactor bugfix |
Bugfix to VPH LBWSG refactor to ensure that LBWSG exposure at birth (rather than the early neonatal exposure) is used for initializing LBWSG exposures in the simulation. Note that this VPH refactor was introduced between models 8.2/8.3 and 9.0 and persisted until this run. |
Baseline |
Default |
|
11.2 |
Update draws for GBD 2021 |
Baseline |
Default |
||
12.0 |
Cap LBWSG RRs and new LNN PAF |
New late neonatal LBWSG PAF calculation, in accordance with vivarium research PR #1681 and subsequent update in PR #1716 |
Baseline |
Default |
|
12.1 |
Preterm prevalence calculation bugfix |
Bugfix to calculation of prevalence of preterm in this equation, to ensure we include categories with an upper bound of 37 weeks |
Baseline |
Default |
|
12.1.1 |
Update LNN LBWSG PAF calculation |
Update to LBWSG PAF calculation for the late neonatal age group. In model 12.0, the PAF calculation for the late neonatal age group did not use the PAF as calculated for the early neonatal age group in the determination of mortality among the early neonatal age group (the PAF using capped and interpolated RRs), as specified in the documentation. This model run will update the LNN LBWSG PAF calculation to utilize the custom calculated ENN LBWSG PAF as specified in the documentation. |
Baseline |
Default |
|
13.0 |
Hemoglobin risk effects on maternal disorders |
Baseline |
Default |
||
13.1 |
Update hemoglobin PAF values and RR handling |
|
Baseline |
Default |
|
13.2 |
Fix LBWSG PAF calculation bugs |
Update to correct bugs in the LBWSG PAF calculation’s implementation of this equation. In earlier model runs, the PAF calculation for late neonates began from age-specific LBWSG prevalence and the mortality-based weighting implemented did not work due to all deaths being excluded by population filters. Instead, in this run, the PAF calculation for late neonates should begin from birth prevalence and properly apply the mortality-based weighting, as documented. |
Baseline |
Default |
|
13.3 |
Update LNN preterm prevalence calculation |
Update to use end-of-ENN LBWSG prevalence for the \(p_\text{preterm}\) for the LNN age group in this equation. Details can be found in the diff of this pull request. |
Baseline |
Default |
|
14.0 |
Wave II antenatal care attendance |
Updates to the antenatal care attendance module |
Baseline |
Default, note that we would like the 4-category ANC attendance variable observed |
|
15.0 |
Delivery facility choice model |
Delivery facility choice model, including updates to the AI Ultrasound module |
Baseline |
Added preterm status and believed preterm status to maternal population observer (#7) |
|
15.1 |
Fix ANC attendance bug and add stratifications |
|
Baseline |
Added preterm status and believed preterm status to maternal population observer (#7) |
|
16.0 |
Wave I antenatal corticosteroids |
Baseline |
Default, note that we would like additional stratifications based on believed gestational age in the maternal population, births, and neonatal burden observers |
||
16.1 |
Facility choice model bugfixes |
Same as previous, but with: believed preterm status added to maternal population (ANC) observer; updated ANC, IFD, and LBWSG propensity values; updates to gestational age estimation error values to match documentation |
Baseline and Ultrasound V&V scenario (scenario #20) |
Default, but add believed preterm stratification to maternal population observer |
|
16.3 (16.2 was skipped because we originally planned to separate the two sets of changes in this model, but did not run them separately) |
Facility choice and neonatal mortality bugfixes |
Same as previous, but with bugfixes. |
Baseline and ultrasound V&V scenario (scenario #20) |
Default, but with noted stratifications added |
|
16.4 |
Fix ACS pipeline and add preterm stratification |
Bugfix to resolve missing values for the |
Baseline |
Default, but with preterm birth status stratification of neonatal deaths observers |
|
16.5 |
Add low hemoglobin RR for depression |
Inclusion of low hemoglobin RR for depressive disorders. No need to actually re-run the model, we just need this RR value active in the interactive context for our custom PAF calculations. |
Baseline |
Default |
|
17.0 |
Oral iron antenatal supplementation (IFA/MMS) |
Oral iron antenatal supplementation (IFA/MMS), including effects on hemoglobin, birth weight, gestational age, and stillbirth. See the hemoglobin module for additional detail. Note this intervention has been implemented in previous models such as nutrition optimization. |
Baseline and MMS scale-up scenarios |
Default, note IFA/MMS coverage added as a stratifying variable to maternal population observer. Also add preterm birth stratification to the births observer. |
|
18.0 |
Anemia screening implementation |
Anemia screening implementation (including hemoglobin and ferritin screenings), see also the hemoglobin module |
Baseline, MMS scale-up, and anemia screening scale-up scenarios |
Default, note hemoglobin and ferritin screening coverage and results added as stratifying variables to maternal population observer |
|
18.1 (not run) |
Oral iron antenatal supplementation (IFA/MMS) bugfixes (interactive sim only) |
Update so that only those who attend ANC are eligible for IFA/MMS; Include corresponding update in the baseline IFA calibration laid out in this PR; Confirm that cat1/cat2 are defined consistently for IFA and MMS coverage and consider updating to covered/uncovered |
No run necessary, all V&V done in the interactive simulation |
N/A |
|
18.2 (not run) |
Oral iron antenatal supplementation (IFA/MMS) bugfixes continued (interactive sim only) |
Updated artifact key for excess shift of IFA on birthweight; Update hemoglobin risk effects to use intervention-affected hemoglobin exposure rather than raw hemoglobin exposure; Update baseline calibration to use coverage among total pop rather than at ANC; Fix common random numbers between scenarios with regard to ANC attendance |
No run necessary, all V&V done in the interactive simulation |
N/A |
|
18.3 |
Fix observer stratifications and multiple bugfixes |
Updated observer requests to avoid >15 stratifications and multiple bugfixes laid out in the outstanding V&V issues table in this PR |
Baseline, MMS scale-up, and anemia screening scale-up scenarios |
Default (note that observer 7 has been broken up into 7a and 7b) |
|
18.4 |
Updated scenarios for GF presentation |
This run to be used for presentation to GF and for record of results pre-GBD 2023 update for comparison |
Scenario numbers 1-9 (all scale-up scenarios for implemented interventions other than misoprostol and hemoglobin-related interventions) |
Default |
|
19.0 (not run) |
GBD 2023 Update part 1 (artifact only) |
Data directly from GBD. See update in draw-level modeling strategy in this PR. Note that as described on the GBD 2023 LBWSG risk exposure document, we will continue to use GBD 2021 data for the LBWSG risk factor exposures and RR values. However, we will still need to re-run the LBWSG RR cap and PAF calculations as they depend on updated mortality risk data. This is an artifact only; the model will not run with this artifact, because it is missing required keys. |
All |
Default |
|
19.0.1 (not run) |
GBD 2023 artifact tweaks and bugfixes |
Sex ratio at birth had been research-team-owned in the past, but this was a simple calculation from a GBD covariate and has been transferred to the engineering side. See this PR for the documentation update. Effect sizes of oral iron supplementation on hemoglobin were erroneously marked as research-team-owned and slated for 19.1; those should be added back unchanged from the 18.X artifacts. The MMS relative risk on stillbirth was incorrect, in both the code and the docs; see this PR for the corrected value. There was some ambiguity about where truncated normal distributions were truncated. This has been clarified in this docs PR. Note this is still just an artifact, and not a model run. |
All |
Default |
|
19.1 |
GBD 2023 Update part 2 |
Data derived from GBD through more complex, research-owned processes. |
All |
Default |
|
19.1.1 |
Fix BEMONC/CEMONC data issue |
Rerun of GBD 2023 Update part 2 with artifacts that resolved data issue in keys dependent on the BEMONC/CEMONC fraction (that were accidentally run on data for the next major model) |
Baseline |
Default |
|
19.1.2 |
Recalculate LBWSG PAFs with capped RRs |
Rerun of previous with LBWSG PAFs recalculated using capped RRs |
Baseline |
Default |
|
20.0 |
In-hospital (CEmONC) delivery estimates from HS team |
In-hospital (CEmONC) delivery estimates from HS team. See PR with diff here |
Baseline |
Default |
|
20.0.1 |
Bugfixes from “In-hospital (CEmONC) delivery estimates from HS team” |
Non-zero neonatal deaths and updated intrapartum azithromycin intervention PAF values |
Baseline |
Default |
|
20.0.2 |
Fix delivery facility model issues |
Baseline |
Default |
||
20.1 |
Sensitivity analysis baseline run without oral iron |
Sensitivity analysis run comparator: same as previous, but without oral iron effects (to sidestep known issues that will be resolved in “remaining pregnancy refactor”), and with more scenarios |
Baseline; AI-assisted ultrasound scale-up; CPAP and ACS scale-up; CPAP, ACS, and AI-ultrasound scale-up |
Default |
|
20.1.1 |
Sensitivity analysis with extreme US error values |
To get an upper bound on the potential impact of AI ultrasound, same as the previous, but set the standard deviation of gestational age error to 70 days for “no ultrasound”, 30 days for “standard ultrasound”, and 2 days for “AI ultrasound”. |
Baseline; AI-assisted ultrasound scale-up; CPAP and ACS scale-up; CPAP, ACS, and AI-ultrasound scale-up |
Default |
|
20.2 |
Sensitivity analysis with lower bound US error values |
To get a lower bound on the potential impact of AI ultrasound for 12/15 Gates meeting, same as the previous, but set the standard deviation of gestational age error to 5 days for “no ultrasound”, 3.5 days for “standard ultrasound”, and 2 days for “AI ultrasound”. |
Baseline; AI-assisted ultrasound scale-up; CPAP and ACS scale-up; CPAP, ACS, and AI-ultrasound scale-up |
Default |
|
20.3 |
Sensitivity analysis with upper bound US error values |
To get an upper bound on the potential impact of AI ultrasound for 12/15 Gates meeting based on additional data seeking, same as the previous, but set the standard deviation of gestational age error to 14 days for “no ultrasound”, 10 days for “standard ultrasound”, and 2 days for “AI ultrasound”. |
Baseline; AI-assisted ultrasound scale-up; CPAP and ACS scale-up; CPAP, ACS, and AI-ultrasound scale-up |
Default |
|
21.0 |
Hemoglobin refactor |
Bringing model up to date with the updated hemoglobin module docs and fixing bugs in “Fix observer stratifications and multiple bugfixes” related to multiple instances of hemoglobin variables that were being inconsistently referenced by different simulation components (See outstanding model verification and validation issues table for full list) |
Baseline, MMS scaleup, and anemia screening scaleup scenarios |
Do not define a measure of “true first trimester hemoglobin exposure” in this version of the model. Otherwise, default. |
|
21.0.1 |
Hemoglobin refactor run correction |
Same as 21.0 but with specific artifact/code discrepancies described in V&V table addressed |
Baseline, MMS scaleup, and anemia screening scaleup scenarios |
Do not define a measure of “true first trimester hemoglobin exposure” in this version of the model. Otherwise, default. |
|
21.1 |
Add dichotomous true hemoglobin output |
Add in dichotomous measure of “true first trimester hemoglobin exposure” for V&V of the hemoglobin screening sensitivity and specificity |
Baseline, MMS scaleup, and anemia screening scaleup scenarios |
Include dichotomous output of true first trimester hemoglobin exposure as a stratification in the anc_hemoglobin observer |
|
21.1.1 |
Hemoglobin refactor: more bugfixes |
Same as previous but with specific bugs described in V&V table addressed |
Baseline, MMS scaleup, and anemia screening scaleup scenarios |
Default |
|
22.0.0 |
Add residual and other maternal disorders |
Inclusion of the residual maternal disorders and abortion/miscarriage/ectopic pregnancy maternal disorders cause models |
Baseline |
Default |
|
22.0.1 |
Residual maternal disorders observer |
Bug fix to include residual maternal disorders in observation |
Baseline |
Default |
|
23.0 |
Remaining pregnancy model refactor |
Specifically with regard to LBWSG exposure. Note that intervention effects on stillbirth are not expected to be resolved in this run. Additionally, include bugfix for inverted baseline anemia screening coverage |
All |
Default |
|
24.0 |
MMS stillbirth effects and GA floors |
|
All, including newly re-added scenarios #12 Azithromycin V&V and #13 Misoprostol V&V (note that scenarios #12 and #13 have been run before, but have been dropped from the branches file) |
Default |
|
22.0.2 |
Residual maternal disorders bugfix |
Fix issue of use of cause ID 1160 rather than cause ID 379 for indirect maternal deaths |
Baseline |
Default |
|
24.1 |
MMS stillbirth effects and GA floors with neonatal deaths bugfix |
Fix bug causing zero neonatal deaths |
All |
Default |
|
24.2 |
MMS stillbirth effects and GA floors with neonatal deaths bugfix and re-generated RR caps |
Same as 24.1 but with re-generated LBWSG RR caps |
All |
Default |
|
24.3 |
GA floor fixes |
Ensure GA floor varies by pregnancy outcome in alignment with docs |
All |
Default |
|
25.0 |
Update SBR to >=24 weeks |
Update to >=24 week stillbirth estimates for SBR. See pull request. Run without oral iron effects (to sidestep known issues and not block this model on “remaining pregnancy refactor”). Note that this should build on top of 20.0.x, not 20.1.x (which was only for sensitivity analysis). |
Baseline |
Default |
|
25.0.1 |
Update SBR to >=24 weeks run bugfixes |
Fix bug in 25.0 causing zero neonatal deaths, ensure run is complete (no failed jobs) |
All |
Default |
None |
26.0 |
IV iron coverage and effect on hemoglobin |
IV iron intervention coverage and effect on hemoglobin. See the hemoglobin module document for more detail. |
Baseline and IV iron scale-up scenarios |
Default, note IV iron coverage as a new stratifying variable to the maternal population observer |
|
24.3 |
GA floor fixes |
Ensure GA floor varies by pregnancy outcome in alignment with docs |
All |
Default |
|
24.4 |
GA floor fixes 2 |
Fix issue with stillbirth GA floor |
All |
Default |
None |
27.0 |
IV iron effects on BW, GA, and stillbirth |
As defined on the IV iron intervention document |
Baseline and IV iron scale-up scenarios |
Default |
|
27.1 |
IV iron neonatal effects bugfixes |
|
Baseline and IV iron scale-up scenarios |
Default |
|
28.0 |
Pakistan fistula update |
Update YLDs due to obstructed labor according to the custom data values specified in this pull request |
Baseline |
Default |
|
28.1 |
Merge 28.0 and 27.1 |
Baseline |
Default |
||
29.0 |
Anemia YLDs and postpartum hemoglobin |
Baseline, MMS scale-up, and IV iron scale-up scenarios |
Default, note new anemia YLD observer |
||
29.1 |
Anemia YLDs sensitivity analysis |
As a sensitivity analysis, disable the effect of oral iron on hemoglobin Note: this model is for sensitivity analysis only and future models should not be based on it. |
Baseline, MMS scale-up, and IV iron scale-up scenarios |
Default |
|
29.2 |
Anemia YLDs sensitivity analysis bugfix |
Fix issue in 29.1 where baseline IFA deletion was still applied to hemoglobin |
Baseline, MMS scale-up, and IV iron scale-up scenarios |
Default |
|
29.0.1 |
Anemia YLDs bugfixes |
Fix issue in 29.0 where anemia YLDs duration for abortion/miscarriage/ectopic pregnancies were longer than the pregnancy; consolidate pregnancy duration and gestational age pipelines |
Baseline, MMS scale-up, and IV iron scale-up scenarios |
Default |
|
29.0.2 |
Anemia YLDs bugfixes 2 |
Fix issue in 29.0.1 where oral iron effects were not being applied to the anemia YLDs calculation |
Baseline, MMS scale-up, and IV iron scale-up scenarios |
Default |
|
29.2.1 |
Merge 29.2 and 29.0.2 |
Merge anemia YLDs bugfixes into sensitivity analysis, and fix a small bug that was causing job failures in the sensitivity analysis run |
Baseline, MMS scale-up, and IV iron scale-up scenarios |
Default |
|
30.0 |
Oral iron GA shift refactor |
Uses different IFA shifts for ANC and non-ANC attendees and recalibrates MMS shifts as described in this PR |
Baseline and MMS scale-up scenarios |
Default |
|
29.0.3 |
Anemia YLDs with pipeline consolidation (PC) |
Consolidate pipelines for pregnancy duration and gestational age, and ensure that the pregnancy duration/gestational age used in the anemia YLDs calculation is consistent with the pregnancy duration/gestational age used in the rest of the model |
Baseline |
Default |
|
29.0.3s |
Anemia YLDs with pipeline consolidation (PC) sensitivity analysis |
Same as 29.0.3, but with oral iron effects on hemoglobin disabled as a sensitivity analysis |
Baseline |
Default |
|
29.0.4 |
Anemia YLDs with stillbirth fix |
Fix issue with stillbirths having negative durations between later ANC and end of pregnancy |
Baseline |
Default |
|
29.0.4s |
Anemia YLDs with stillbirth fix sensitivity analysis |
Same as 29.0.4, but with oral iron effects on hemoglobin disabled as a sensitivity analysis |
Baseline |
Default |
|
29.0.5 |
Anemia YLDs with person-time observer fix |
Fix issue with anemia person-time observer where it did not sum across simulants in the postpartum timestep |
Baseline |
Default |
|
29.0.5s |
Anemia YLDs with person-time observer fix sensitivity analysis |
Same as 29.0.5, but with oral iron effects on hemoglobin disabled as a sensitivity analysis |
Baseline |
Default |
|
31.0 |
Update hemoglobin exposure to release ID 33 |
Merges 29.0.5 and 30.0, and updates hemoglobin exposure to release ID 33 |
Baseline |
Default |
|
31.0s |
Update hemoglobin exposure to release ID 33 sensitivity analysis |
Same as 31.0, but with oral iron effects on hemoglobin disabled as a sensitivity analysis |
Baseline |
Default |
|
Larger run for neonatal mortality V&V |
Includes “neonatal all-cause mortality risk”, “neonatal cause-specific mortality risks”, and “impossible neonatal CSMRisk” observers. |
Baseline |
|
Oral iron GA shift optimization and facility choice model interaction resolution? |
|
Update hemoglobin effects |
As defined on the hemoglobin risk effects document (Custom PAFs and neonatal sepsis effects have yet to be calculated for GBD 2023): Updated custom PAF values for maternal hemorrhage and maternal sepsis outcomes (paired with existing implementation of GBD RRs); New risk effect (using GBD RRs and custom PAFs) for depressive disorders; New risk effect (using custom RRs and PAFs) for neonatal sepsis |
Baseline and IV iron scale-up scenarios |
Default |
RT-owned data generation that is blocked by Separate LBWSG affected causes run |
|
Effects of maternal disorders on postpartum hemoglobin |
Effects of maternal hemorrhage (and possibly maternal sepsis) on postpartum hemoglobin. Model run is blocked by |
Baseline, MMS scale-up, and IV iron scale-up scenarios |
Default |
Research tickets to update maternal hemorrhage risk effect docs to GBD 2023 and consider adding risk effect for maternal sepsis |
|
Separate LBWSG affected causes |
Update neonatal mortality model to treat LBWSG-affected and -unaffected causes differently in accordance with this pull request |
All |
Default |
Larger run for neonatal mortality V&V run |
|
Trimester-specific ultrasound |
Update ultrasound model to include gestational age estimation error specific to timing of ultrasound in addition to ultrasound type. See the ultrasound module document for details |
All |
Default, note that observed value for ultrasound is now “ultrasound summary” rather than “ultrasound type” with this update |
Updated data values for trimester-specific GA error values, updated facility choice model and values that reflect the updated GA error values, Remaining pregnancy model refactor run |
Note
Some of the notebook URLs for the older runs might be out-of-date. If you click one of these links and it gives you a 404 error, add to your URL /old_vnv_notebooks/ after verification_and_validation, and that should take you to the right place!
Model number |
Short name |
V&V plan |
V&V summary |
Link to notebook |
|---|---|---|---|---|
1 |
Wave I Pregnancy V&V |
|
All checks passed except last one; RT is updating our observer output requests to add an observer for pregnant person age. |
|
2 |
Wave I Maternal disorders V&V |
|
All checks passed except error found in GBD 2021 for Pakistan fistula modeling - need to update the artifact for Pakistan OL prevalence values from GBD 2021 to GBD 2023. Did not explicitly check YLLs yet. |
|
3 |
Wave I Neonatal disorders V&V |
|
Found an error in LBWSG distribution in artifact, which might be the cause of some of the other checks that weren’t passing, including the ACMR for the late neonatal group and the CSMR for preterm |
|
3.1 |
Wave I Neonatal disorders V&V with correct LBWSG distribution |
Validate LBWSG exposure distribution |
LBWSG distributions in artifact, GBD, and simulation are now matching, but preterm deaths still look too low in the simulation |
|
3.2 |
Wave I Neonatal disorders V&V with LBWSG component removed |
Validate all-cause mortality for early and late neonatal age groups with LBWSG component removed |
Early neonatal mortality is still being overestimated in the simulation |
|
3.3 |
Wave I Neonatal disorders V&V with early NN observer bugfix |
|
Early neonatal mortality is validating now! Note: Ali noticed in the LBWSG interactive sim that the state table and pipeline values for LBWSG exposure don’t match, but engineers confirmed this is okay, the pipeline values refresh after being recorded in the state table (and then are not used again). |
|
4.1 |
Wave I CPAP |
Validate RR of CPAP on RDS preterm (and confirm other causes are unchanged) |
Cannot validate, need observer with counts per facility type |
|
4.2 |
Wave I CPAP with observer for counts per facility type |
Validate RR of CPAP on RDS preterm (and confirm other causes are unchanged) |
Cannot validate, need to add delivery facility column in births observer and stratification for CPAP availability |
|
4.3 |
Wave I CPAP with addition of a delivery facility column |
Validate RR of CPAP on RDS preterm (and confirm other causes are unchanged) |
Not validating, need to update how we determine which delivery facility type a simulant will go to |
|
4.4 |
Wave I CPAP with updated facility determination |
Validate RR of CPAP on RDS preterm (and confirm other causes are unchanged) |
Not validating, we are seeing negative mortality rates for Other causes |
|
4.5 |
Wave I CPAP with mortality bugfix |
Validate RR of CPAP on RDS preterm (and confirm other causes are unchanged) |
CSMRs and ACMR are all validating now, with the bugfix to adjust all negative values to 0 and rescale the rest of the RRs to add up to 1 |
|
4.7 |
Correct pregnancy duration for abortion/miscarriage/ectopic pregnancies |
Validate abortion/miscarriage/ectopic pregnancy duration is between 6 and 24 weeks and uniformly distributed. |
Validated for all 3 locations |
|
5.0 |
Wave I neonatal antibiotics with scale-up scenarios |
Validate RR of antibiotics on sepsis (and confirm other causes are unchanged) |
Everything is validating - RR on sepsis aligns with expected value; other causes, non-RDS preterm, and encephalopathy all have the expected RRs of 1 from antibiotics. There’s an RR of 0.78 for antibiotics on preterm with RDS, but we confirmed that when we group this by facility type, there is the expected RR of 1. This is because the probability of a simulant receiving CPAP and the probability of receiving antibiotics are not independent (both related to facility choice). |
|
5.1 |
Wave I neonatal antibiotics refactor |
Validate maternal and neonatal disorders and intervention effect sizes after refactor |
Everything is validating! We noticed the maternal disorders incidence parquet files were mislabeled, the fix for that has already been implemented. |
|
6.0 |
Wave I neonatal probiotics with scale-up scenarios |
Validate coverage, RR of probiotics on sepsis (and confirm other causes are unchanged) |
Neonatal ACMR looks off, residuals have gotten increasingly worse with additional interventions |
|
6.0.1 |
Wave I neonatal disorders ACMR 200k |
Validate neonatal disorders ACMR with 200k population without interventions |
Used the attached notebook and spreadsheet to figure out which runs were validating with ACMR and which were not |
|
6.0.2 |
Wave I neonatal disorders ACMR 2M |
Validate neonatal disorders ACMR in baseline scenario with 2 million population |
Used the attached notebook and spreadsheet to figure out which runs were validating with ACMR and which were not |
|
6.0.3 |
Wave I neonatal disorders ACMR with rate conversion |
Validate neonatal disorders ACMR when reverting the rate to probability conversion for mortality rates when choosing when neonates die |
Used the attached notebook and spreadsheet to figure out which runs were validating with ACMR and which were not |
|
6.0.4 |
Wave I neonatal disorders ACMR with raw CSMR |
Validate neonatal disorders ACMR when using raw CSMRs for the non-preterm neonatal causes, removed LBWSG RRs on those neonatal causes |
Used the attached notebook and spreadsheet to figure out which runs were validating with ACMR and which were not |
|
6.1 |
LBWSG PAF changes for Ethiopia |
Check ENN mortality ratio compared to GBD |
Neonatal mortality ratios are now slightly underestimated (rather than the previous overestimation). Note that calculation of the mortality ratio of the LNN age group has been updated in this notebook to be [deaths in LNN age group] / [population at the start of the LNN age group], rather than a denominator of live births so that LNN mortality is not dependent on ENN mortality. |
|
6.2 |
Exponential rate-to-probability conversion |
Check ENN mortality ratio compared to GBD |
Neonatal mortality ratios are now dramatically overestimated. Note that while the birth observer has changed between models 6.1 and 6.2, it has been verified that birth counts do not vary between these runs and that greater death count values are driving the difference between neonatal mortality ratios in 6.1 and 6.2 |
|
6.2.1 |
Fix rate-to-probability equation |
Check ENN mortality ratio compared to GBD, check that birth observer is recording abortion/miscarriage/ectopic pregnancies |
|
|
6.3 |
ENN LBWSG PAF using ENN exposure |
Check ENN mortality ratio compared to GBD and models 6.1-6.4 |
Mortality is slightly overestimated. It appears that overestimation in 6.3 is slightly larger in magnitude than the underestimation of 6.1. |
|
6.4 |
Revert to linear rate-to-probability |
Check ENN mortality ratio compared to GBD and models 6.1-6.4 |
Mortality is overestimated to a degree greater than 6.3 |
|
6.5 |
Add maternal population observer |
|
|
|
7.0 |
Wave I neonatal probiotics with effective coverage |
|
|
|
7.0.1 |
Add preterm stratification and fix observers |
|
All specified V&V criteria looks great! Did notice that CPAP relative risk in artifact is a point value despite having uncertainty specified in documentation. |
|
7.0.2 |
Update preterm birth parameter |
Check that preterm birth mortality is as expected: we should change from a slight overestimation to a slight underestimation. A slight underestimation is expected due to known mortality probabilities greater than 1, which will be addressed in future model runs. |
The overestimation of preterm birth mortality is of lower magnitude than in 7.0.1, indicating that the update of the preterm prevalence term improved the model. However, preterm birth mortality remains slightly overestimated on average rather than the expected slight underestimation. |
|
7.1 |
Update neonatal mortality to risks |
|
|
|
7.1.1 |
Add CPAP parameter uncertainty |
|
All looks good except the artifact values for the CPAP relative risk are not quite as expected due to issue raised in this comment |
|
8.0 |
Wave I azithromycin |
|
|
|
8.1 |
Cap LBWSG RRs |
|
|
|
8.2 |
Update neonatal probiotics effect size |
|
All looks good! |
|
8.3 |
Update antibiotics modeling strategy |
|
All looks good, except antibiotics coverage is not being scaled up among those who deliver at home as it should be |
|
9.0 |
Wave I misoprostol |
|
|
|
9.1 |
Antibiotics bugfix for home deliveries |
|
|
|
9.2 |
Larger population for obstructed labor V&V |
|
Same conclusions as 9.0 |
|
9.3 |
Additional stratifications for intrapartum V&V |
Confirm intrapartum interventions are meeting V&V criteria |
Intrapartum intervention coverage and effects are looking just as expected :) |
|
10.0 |
Add postpartum depression |
|
All looks great! |
|
10.1 |
Remove LBWSG effect on neonatal encephalopathy |
Check if cause-specific neonatal mortality validates |
|
|
10.2 |
Same as “Add postpartum depression” with scenario #13 |
Confirm baseline mortality is as expected, scenario-specific intervention coverage is as expected |
Looks as expected (including persistent NN mortality underestimation that arose in model 9.0) |
|
11.0 |
Add hemoglobin risk exposure model |
|
All looks good! However, we are not using the draw numbers pre-specified in this PR. The draws that have been run include duplicate hemoglobin exposure values. |
|
11.1 |
VPH LBWSG refactor bugfix |
|
Looks good! |
|
11.2 |
Update draws for GBD 2021 |
Check that draw numbers have been updated |
Looks good! |
|
12.0 |
Cap LBWSG RRs and new LNN PAF |
|
|
|
12.1 |
Preterm prevalence calculation bugfix |
|
Neonatal cause-specific mortality risks match expectation for both preterm and non-preterm causes in the early neonatal period. We appear to be systematically underestimating preterm CSMRisks in the late neonatal period. |
|
12.1.1 |
Update LNN LBWSG PAF calculation |
|
|
|
13.0 |
Hemoglobin risk effects on maternal disorders |
|
|
|
13.1 |
Update hemoglobin PAF values and RR handling |
|
|
|
13.2 |
Fix LBWSG PAF calculation bugs |
|
|
|
13.3 |
Update LNN preterm prevalence calculation |
|
|
Model 13.3 neonatal checks Model 13.3 interactive sim neonatal mortality checks |
14.0 |
Wave II antenatal care attendance |
|
All V&V criteria met! |
|
15.0 |
Delivery facility choice model |
Checks using observer outputs:
Checks using interactive sim:
|
Measures meeting V&V criteria:
V&V issues:
Not able to be checked:
|
|
15.1 |
Fix ANC attendance bug and add stratifications |
Same as 15.0 |
Measures meeting V&V criteria:
Known issues:
V&V targets not met: (thought to be related to “known issues”)
|
|
16.0 |
Wave I antenatal corticosteroids |
|
|
|
16.1 |
Facility choice model bugfixes |
|
All V&V criteria met except for:
(Note that scenario #20 was not included in this run, so related V&V will be performed in 16.3 instead) |
|
16.3 |
Facility choice and neonatal mortality bugfixes |
For facility choice: Same as 16.1 and 16.0 For neonatal mortality:
|
|
|
16.4 |
Fix ACS pipeline and add preterm stratification |
Same as 16.3 |
|
|
17.0 |
Oral iron antenatal supplementation (IFA/MMS) |
|
|
|
18.0 |
Anemia screening implementation |
|
|
Interactive simulation notebook for model 18.0 found here. Note that simulation results for this run were not generated due to too many observer stratifications. |
18.1 |
Oral iron antenatal supplementation (IFA/MMS) bugfixes (interactive sim only) |
|
|
|
18.2 |
Oral iron antenatal supplementation (IFA/MMS) bugfixes continued (interactive sim only) |
Same as 17.0 |
|
|
18.3 |
Fix observer stratifications and multiple bugfixes |
Same as 17.0 and 18.0 |
Meeting the following criteria:
Partially meeting the following criteria:
Not meeting the following criteria:
|
|
19.1 |
GBD 2023 Update part 2 |
|
|
|
19.1.1 |
Fix BEMONC/CEMONC data issue |
Same as 19.1 and confirm specific issues from 19.1 are resolved |
|
Model 19.1.1 V&V notebooks Model 19.1.1 interactive sim neonatal mortality V&V notebook |
19.1.2 |
Recalculate LBWSG PAFs with capped RRs |
Same as 19.1.1 and confirm specific issues from 19.1.1 are resolved |
|
|
20.0 |
In-hospital (CEmONC) delivery estimates from HS team |
|
|
|
20.0.1 |
Bugfixes from In-hospital (CEmONC) delivery estimates from HS team |
|
|
|
20.0.2 |
Fix delivery facility model issues |
|
|
|
20.1 |
Sensitivity analysis baseline run without oral iron |
|
Outputs looks as expected! |
|
20.1.1 |
Sensitivity analysis with extreme US error values |
|
Outputs look as expected! |
|
20.2 |
Sensitivity analysis with lower bound US error values |
|
Outputs look as expected! |
|
20.3 |
Sensitivity analysis with upper bound US error values |
|
Outputs look as expected! |
|
21.0 |
Hemoglobin refactor |
Using simulation outputs:
In the interactive simulation:
Note that the effects of hemoglobin on LBWSG exposure or pregnancy outcome are not yet expected to meet V&V criteria. |
Could not run facility choice V&V because PR exposing the V&V targets had not been integrated into the branch this was run from. On further inspection, artifact had been based on 20.1.1 (which was for sensitivity analysis only). |
|
21.0.1 |
Hemoglobin refactor run correction |
Same as 21.0 |
21.0.1 was only partially V&Ved, checking criteria that did not require an interactive sim. This partial V&V surfaced two bugs: believed preterm was always false, and ACS eligibility was always false. |
|
21.1 |
Add dichotomous true hemoglobin output |
|
Not V&Ved before next model was run. |
|
21.1.1 |
Hemoglobin refactor: more bug fixes |
Combination of 21.0 and 21.1. |
|
|
22.0.0 |
Add residual and other maternal disorders |
|
Residual maternal disorders not included in observation |
No notebooks due to lack of observers, see 22.0.1 |
22.0.1 |
Residual maternal disorders observer |
Same as 22.0.0 |
Everything matches, except that cause ID 1160 rather than 379 was included in “residual maternal disorders” |
|
22.0.2 |
Residual maternal disorders bugfix |
Same as 22.0.0 |
Everything matches |
|
23.0 |
Remaining pregnancy model refactor |
In the simulation outputs:
In the interactive simulation:
|
|
|
24.0 |
MMS stillbirth effects and GA floors |
|
|
V&V notebooks included in this PR |
24.1 |
MMS stillbirth effects and GA floors with neonatal deaths bugfix |
Same as 24.0 |
|
V&V notebooks included in this PR |
24.2 |
MMS stillbirth effects and GA floors with neonatal deaths bugfix and re-generated RR caps |
Same as 24.0 |
|
V&V notebooks included in this PR |
24.3 |
Same as 24.0 |
|
V&V notebooks included in this PR |
|
24.4 |
GA floor fixes 2 |
In the interactive simulation, confirm that minimum gestational age values stratified by pregnancy outcome match floors documented, minus baseline IFA calibration shift |
|
V&V notebooks included in this PR |
25.0 |
Update SBR to >=24 weeks |
|
|
|
25.0.1 |
Update SBR to >=24 weeks run bugfixes |
Same as 25.0, plus: * Neonatal mortality matches expectation * Regression tests pass (no CRN issues due to failed jobs) |
|
|
26.0 |
IV iron coverage and effect on hemoglobin |
|
|
|
27.0 |
IV iron effects on BW, GA, and stillbirth |
|
|
|
27.1 |
IV iron effects on BW, GA, and stillbirth bugfixes |
Same as 27.0, with issues noted there fixed |
All checks passed |
Model 27.1 V&V notebooks are spread across two PRs: here and here |
28.0 |
Pakistan fistula update |
|
All checks passed |
|
28.1 |
Merge 28.0 and 27.1 |
Regression testing |
All checks passed |
|
29.0 |
Anemia YLDs |
|
|
|
29.1 |
Anemia YLDs sensitivity analysis |
|
|
|
29.2 |
Anemia YLDs sensitivity analysis bugfix |
Same as 29.1 |
|
|
29.0.1 |
Anemia YLDs bugfixes |
Same as 29.0, and that issues noticed in 29.2 are resolved |
Results indicate that oral iron effects on gestational age are not being applied |
|
29.0.2 |
Anemia YLDs bugfixes 2 |
Same as 29.0.1, and confirm that oral iron effects on gestational age are being applied |
Round 1:
Round 2:
|
|
29.2.1 |
Merge 29.2 and 29.0.2 |
Same as 29.2 |
|
|
30.0 |
Oral iron GA shift refactor |
|
|
|
29.0.3 |
Anemia YLDs with pipeline consolidation (PC) |
Same as 29.0.2 |
|
|
29.0.3s (note that this was based on 29.2.1 in addition to 29.0.3; this was a numbering error) |
Anemia YLDs with pipeline consolidation (PC) sensitivity analysis |
Same as 29.2.1 |
YLD rates and prevalence rates still look high, in addition to YLDs per pregnancy, but they are lower than in 29.0.3 |
|
29.0.4 |
Anemia YLDs with stillbirth fix |
Same as 29.0.3 |
|
|
29.0.4s |
Anemia YLDs with stillbirth fix sensitivity analysis |
Same as 29.0.3s |
|
|
29.0.5 |
Anemia YLDs with person-time observer fix |
Same as 29.0.4 |
|
|
29.0.5s |
Anemia YLDs with person-time observer fix sensitivity analysis |
Same as 29.0.4s |
|
|
31.0 |
Update hemoglobin exposure to release ID 33 |
Confirm that hemoglobin checks continue to pass, with validation targets updated to release ID 33 |
|
|
31.0s |
Update hemoglobin exposure to release ID 33 sensitivity analysis |
Same as 29.0.5s, but with hemoglobin exposure updated to release ID 33 |
|
|
Larger run for neonatal mortality V&V |
Confirm expected rates of cause-specific and overall maternal disorders causes |
|||
Update hemoglobin effects |
|
|||
Effects of maternal disorders on postpartum hemoglobin |
|
|||
Separate LBWSG affected causes |
|
|||
Trimester-specific ultrasound |
|
Issue |
Explanation |
Action plan |
Timeline |
|---|---|---|---|
Unnecessary data assigned to abortion/miscarriage/ectopic pregnancy in the interactive simulation |
Including gestational age/birth weight exposures as well as neonatal mortality risk |
Engineers to update at their convenience |
TBD |
Overestimating proportion of believed term given preterm fraction |
Originally thought to be related to failure to account for additional correlation induced by the baseline IFA calibration with respect to gestational age, but this has not been resolved by model 30.0 updates to the oral iron GA shift implementation. New theory is that our calibration causes mean gestational age to match, but not the proportion of preterm. |
Research to investigate <https://jira.ihme.washington.edu/browse/SSCI-2614> |
TBD |
Early neonatal other causes mortality risk in Pakistan overestimated |
Unknown |
Assess whether this is due to the negative other causes mortality rate issue in the neonatal mortality V&V run |
Neonatal mortality V&V run |
Abortion/miscarriage/ectopic pregnancies have non-null values for preterm birth, believed preterm, ACS eligibility in observed simulation results. While we can filter these results out, it presents opportunity for error in analyzing results and these values should be updated to N/A for partial term pregnancies |
Partial term pregnancies are assigned LBWSG exposures in the interactive sim and therefore are observed for these outputs |
Engineers to update at the same time as implementing observer revamp (not yet documented as of 12/18/25) |
TBD |
There is zero coverage of “ACS availability” among stillbirths even though stillbirths should be eligible and covered by this intervention. |
Likely a result of there being null coverage for CPAP availability for stillbirths (because they are not alive to receive CPAP). However, stillbirths should receive ACS coverage if they are in the relevant believed gestational age range and delivering in a facility that has CPAP access. |
Wait until we split stillbirths into antepartum and intrapartum before we address this issue, as only intrapartum stillbirths should receive ACS coverage (?) |
TBD |
Ali’s documentation issue resulted in known issues with ferritin data used for implementation of anemia screening model |
Either update to strategy outlined in this PR or an alternative strategy using PRISMA data shared by the Gates foundation |
Will decide how to proceed when we receive PRISMA data |
|
Unable to verify ferritin screening results among those who are not anemic according to hemoglobin level at the time of testing |
Re-address when we update ferritin exposure model |
Maintain existing behavior for now |
TBD |
Current implementation is based off of an adaptation of the assumptions used in the GBD 2021 major depressive disorders cause model |
We will need to either (1) update our model to be in line with the GBD 2023 model and consider updating our PAF calculation strategy as described in this ticket, or (2) update to the extra-GBD data on peripartum depression obtained from the mental disorders modelers |
Will decide how to proceed after discussing with the mental disorders modelers |
|
Miscalibration of maternal sepsis incidence rates, particularly for Nigeria |
Thought to be due to using the fatal PAF from GBD applied to incidence and/or the location-aggregated PAF for our modeled locations which are not most detailed locations |
Update to custom-calculated PAF and reassess |
TBD |
Late neonatal mortality due to preterm birth slightly underestimated and other-causes mortality may be slightly overestimated (though within 10%) |
Unknown – possibly related to negative other causes mortality in Pakistan and Nigeria. |
Neonatal mortality observers |
Larger run for neonatal mortality V&V |
Late neonatal mortality due to preterm birth with RDS slightly (~2%) underestimated |
The PAF of ACS and CPAP on preterm birth with RDS CSMRisk is calculated with delivery facility proportions at birth, not at 7 days |
Accept this limitation, until/unless there are other reasons to revamp PAF calculation, since this would require many components not currently present in PAF sim |
N/A |
Early neonatal mortality due to preterm birth with RDS slightly (~1.5%) overestimated and late neonatal mortality slightly (~0.75%) underestimated across all causes |
The PAF of ACS and CPAP on preterm birth with RDS CSMRisk is calculated without accounting for correlation between LBWSG and CPAP/ACS, which is induced by the facility choice model. The miscalibration in ENN causes miscalibration of LBWSG exposure for LNN. |
Accept these limitations, until/unless there are other reasons to revamp PAF calculation, since this would require many components not currently present in PAF sim. Note that the LNN limitation stacks with the previous limitation for preterm birth with RDS to result in a nearly 3% underestimate in that LNN CSMRisk. |
N/A |
In GBD 2023 data for Pakistan the mortality values for the abortion and miscarriage cause (c_995) are very small (nearly the lowest of any national location globally), causing unexpectedly low YLLs (~100 times fewer than India) |
Possible issue with ST-GPR model reacting to an all-zero datapoint added in GBD 2023 for Pakistan |
Determine cause of issue with GBD modeling team, decide whether to leave as-is or use a proxy location |
TBD |
LBWSG exposures change between scenarios for simulants whose birth outcome changes between scenarios |
Due to different exposure distributions used for stillbirths vs. livebirths, given the different floors. |
Accept this limitation |
N/A |
Severe anemia underestimated due to underestimate of hemoglobin exposure standard deviation for aggregated locations |
Hemoglobin exposure SDs for aggregated locations have been generated by central machinery as a population-weighted mean of the SDs for the most detailed locations, which would only be correct if the mean exposure were the same across the most detailed locations. |
GBD anemia team to update the database and/or provide a flat file for these SDs |
N/A |
Moderate anemia overestimated due to IFA delays |
We delete baseline IFA from all simulants and only add back the impact on hemoglobin when they receive IFA at ANC. This inflates anemia prevalence vs the GBD hemoglobin distribution. |
Accept this limitation for now; if we revisit the baseline IFA deletion in the future, we can reassess this issue |
N/A |
6.0 Limitations
Unclear if we will be able to include upstream factors, but these are likely correlated with many things such as ANC visit rate, care available, or even outcome rates
We currently do not consider the potential impact of pregnancy outcome, mode of delivery, or preterm status on postpartum depression incidence (although we do model the impact of hemoglobin on PPD incidence). This may cause us to underestimate the impact of interventions that work through these mechanisms on postpartum depression burden.
Factors such as birth asphyxia have been shown to predisopose infants to infection which can result in sepsis [Tikmani-et-al-2016]. We do not model a relationship between birth asphyxia and sepsis, so we do not capture any indirect effects of interventions to reduce birth asphyxia (like cesarean sections) on sepsis as mediated through reductions in birth asphyxia.
We assume that all maternal deaths occur at the conclusion of the intrapartum period and prior to the start of the postpartum period. Therefore, we assume it is not possible for any simulants who die of a maternal disorder to experience postpartum YLDs (such as those due to postpartum depression and/or postpartum anemia). However, this may be possible in reality, particularly for those who die of “late maternal deaths.”
We track certain outcomes among abortion/miscarriage/ectopic pregnancies in this model, including first trimester ANC attendance and associated interventions, anemia YLDs, and postpartum depression. However, these pregnancies are not given special consideration other than their premature end and we do not consider how this population may differ from pregnancies that end in live or still births in terms of their ANC attendance rates or other attributes beyond maternal disorders burden. Additionally, we do not model any variation in these attributes by subtype (abortion vs. miscarriage vs. ectopic pregnancy), despite there being expected differences in behavior between these groups.
We do not model an underlying correlation between hemoglobin exposure and stillbirth rates, despite evidence that such an association exists. Therefore, our IV iron intervention model, which is targeted to those with low hemoglobin, will be misaligned with respect to the stillbirth rate among the IV iron intervention target population.
We could use the GBD risk effects between hemoglobin and stillbirth to model baseline correlation only and not model updates in stillbirth rates in response to changes in hemoglobin exposure to address this limitation (as these effects are captured in the impact of the hemoglobin-affecting interventions IV Iron and IFA/MMS already). However, this model upgrade is not highest priority. See this backlog JIRA ticket #2343
[Finkelstein-et-al-2024] primarily includes RCTs from high-income countries, so the effect size for IFA on maternal hemoglobin may be overestimated for Sub-Saharan African countries (including Ethiopia and Nigeria) with typically higher rates of non-iron deficiency anemias.
We do not currently model an oral iron treatment intervention, despite the recommendation by WHO to treat all pregnancies with IFA and all anemic pregnancies with oral iron treatment (essentially a higher dosage of iron than IFA). We have a JIRA ticket to add this intervention in the future, but it is not currently prioritized. There are some individuals who receive oral iron treatment at baseline and we want to model the replacement of that oral iron treatment with IV iron treatment. However, we are not subtracting out the effect of oral iron treatment prior to modeling the scale up of IV iron treatment. This will result in the following limitations:
For individuals who received oral iron treatment at baseline and still remain eligible for IV iron following their oral iron treatment: we will double count the impact of the interventions, thus overestimating the potential scale-up of IV iron.
For individuals who received oral iron treatment at baseline and no longer remain eligible for IV iron following their oral iron treatment: there will be essentially no difference in impact for this population given that IV and oral iron treatment have basically equal impacts, however, we will be underestimating the size of the population requiring treatment and therefore underestimating the cost required to achieve the health outcomes seen in these scenarios.
7.0 References/Other
See the “(“Darmstadt 2011” reference in: Tikmani SS, Muhammad AA, Shafiq Y, Shah S, Kumar N, Ahmed I, Azam I, Pasha O, Zaidi AK. Ambulatory Treatment of Fast Breathing in Young Infants Aged <60 Days: A Double-Blind, Randomized, Placebo-Controlled Equivalence Trial in Low-Income Settlements of Karachi. Clin Infect Dis. 2017 Jan 15;64(2):184-189. doi: 10.1093/cid/ciw690. Epub 2016 Oct 19. PMID: 27941119; PMCID: PMC5853586.
Note
This concept model was reorganized in April 2025 after much of wave I had been implemented. A record of the PRs for this reorganization are listed below:
Pregnancy component reorganization: https://github.com/ihmeuw/vivarium_research/pull/1615
Intrapartum component reorganization: https://github.com/ihmeuw/vivarium_research/pull/1617
Neonatal component reorganization: https://github.com/ihmeuw/vivarium_research/pull/1619