GBD 2023 Maternal Hemorrhage Risk Effects
Risk Overview
This page describes the Vivarium modeling strategy for risk effects. For a description of Vivarium modeling strategy for risk exposure (in this case a cause model document), see the maternal hemorrhage page.
GBD 2023 Modeling Strategy
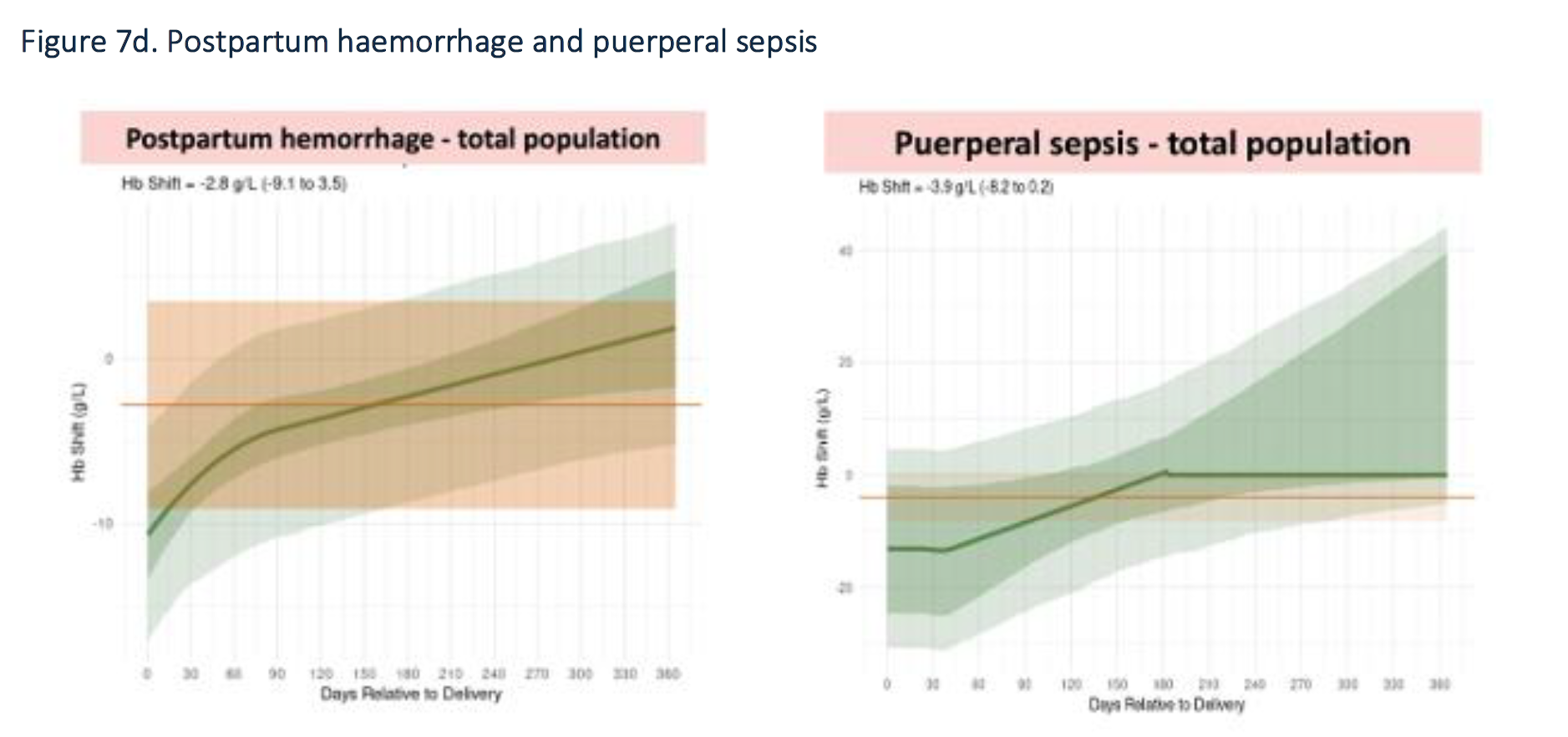
GBD does not explicitly model maternal hemorrhage as a risk factor. However, GBD models a hemoglobin shift associated with maternal hemorrhage in the anemia causal attribution process. Newly for GBD 2023, this hemoglobin shift is time-dependent (by days relative to delivery) and is informed by an observational analysis of US MarketScan data. The graph below is from the nonfatal methods appendix. Note that it compares postpartum people with postpartum hemorrhage to postpartum people without postpartum hemorrhage, so the hemoglobin shift values are in addition to the hemoglobin changes associated with pregnancy itself.
Vivarium Modeling Strategy
Outcome |
Outcome type |
Outcome ID |
Affected measure |
Note |
|---|---|---|---|---|
Hemoglobin concentration during the first six weeks after the end of pregnancy |
Risk exposure |
376 |
Hemoglobin concentration |
During the first six weeks after the end of pregnancy only |
Hemoglobin concentration between 6 weeks and 9 months after the end of pregnancy |
Modelable entity |
27596 |
Hemoglobin concentration |
During the period from 6 weeks to 9 months after the end of pregnancy only; this is the “non-pregnant” hemoglobin distribution |
Hemoglobin effects after the end of pregnancy
For simulants who experience an incident case of maternal hemorrhage (moderate or severe) as determined in the maternal hemorrhage page,
we will decrease the simulant’s hemoglobin concentration during two distinct time periods: the first six weeks after the end of pregnancy and the period from 6 weeks to 9 months after the end of pregnancy.
We chose the 9 month cutoff because this is the point at which the GBD 2023 shift curve for postpartum hemorrhage returns to approximately 0, indicating no difference in hemoglobin concentration between those with and without postpartum hemorrhage;
we do not find the positive shifts after 9 months postpartum to be biologically plausible and therefore do not apply a positive hemoglobin shift after 9 months postpartum.
The magnitude of the decrease will be determined by the maternal hemorrhage hemoglobin effect estimated in GBD 2023,
which is time-dependent by days relative to delivery.
For each time period, we take an average of the continuous hemoglobin shift curve across that time period to determine the hemoglobin shift value applied during that time period.
The curve was provided to us by the anemia modelers at /mnt/team/anemia/pub/emotive/mat_hem/, specifically in the pred_data.csv file in that directory.
We will copy this file to our simulation repository for tracking purposes.
Validation and Verification Criteria
The hemoglobin concentration stratified by maternal hemorrhage incidence (also stratified by anemia status in pregnancy to avoid confounding by this factor) should differ by the magnitude of the maternal hemorrhage hemoglobin effect, in each post-pregnancy time step.
Assumptions and Limitations
This modeling strategy does not consider that the impact of maternal hemorrhage is already reflected in the pregnancy adjustment factor used for the hemoglobin model and therefore we may slightly underestimate hemoglobin concentration (and therefore overestimate anemia prevalence) on average during the pregnancy and lactation period by applying an additional negative hemoglobin shift associated with maternal hemorrhage.
This modeling strategy applies an average hemoglobin shift for all incident maternal hemorrhage cases during the first six weeks after the end of pregnancy and an average hemoglobin shift for all incident maternal hemorrhage cases during the period from 6 weeks to 9 months after the end of pregnancy, which does not capture the continuous nature of hemoglobin changes over time. The continuous curves could result in simulants moving between several anemia categories over the course of the postpartum period, which is not captured here. This also means that the (relatively arbitrary) choices of durations of these periods are impactful.
We currently apply the same hemoglobin shift for all incident maternal hemorrhage cases, but in reality the hemoglobin shift likely differs by severity of maternal hemorrhage (moderate vs severe), which is not captured here. We may address this in the future based on our approach in the previous maternal hemorrhage effects model.
The GBD shift is derived from data about postpartum hemorrhage only, and we are applying it to all maternal hemorrhage cases (including antepartum hemorrhage), which may not be appropriate. We may address this in the future by using GBD crosswalk models to split antepartum vs postpartum hemorrhage cases.
The GBD shift is derived from USA MarketScan data, which may not be generalizable to other locations. In particular, treatment in the USA may be more effective at mitigating the hemoglobin shift associated with maternal hemorrhage than treatment in other locations, which could lead to an underestimation of the hemoglobin shift in locations with less effective treatment. Additionally, the USA has more access to ultrasound than our modeled locations which could lead to better prevention.
Todo
Consider a modeling strategy that calibrates the pregnancy-specific hemoglobin exposure to the baseline level of maternal hemorrhage in the population